Granules India receives USFDA approval for Trazodone Hydrochloride Tablets
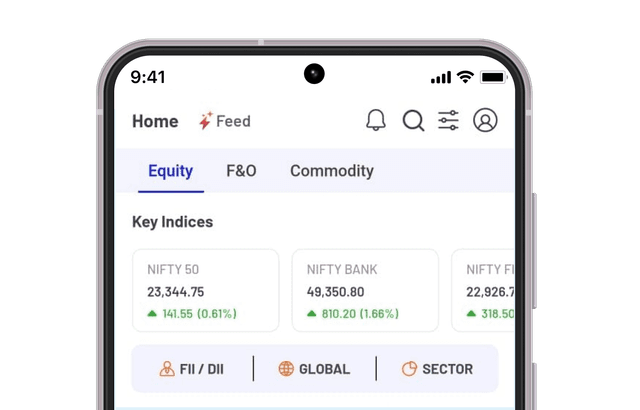
Granules India announced today that the US Food & Drug Administration (US FDA) has approved its Abbreviated New Drug Application (ANDA) for Trazodone Hydrochloride Tablets USP, 50 mg, 100 mg, 150 mg, and 300 mg. It is bioequivalent and therapeutically equivalent to the reference listed drug, Desyrel Tablets, 50 mg, 100 mg, 150 mg, and 300 mg, of Pragma Pharmaceuticals, LLC.
Trazodone tablets are indicated for the treatment of major depressive disorder in adults.
Dr. Krishna Prasad Chigurupati, Chairman and Managing Director, Granules India Limited said, “We have received the approval within 10 months of filing the application, which is a testament to our strong R&D capabilities. The product will be launched within this quarter and the market share will grow steadily over the next few quarters.”
0 mins read . Dec 18, 2024

0 mins read . Dec 18, 2024

0 mins read . Dec 18, 2024