Caplin Steriles receives USFDA approval for Milrinone Lactate in 5% Dextrose Injection infusion bags
Caplin Steriles (Caplin), a subsidiary company of Caplin Point Laboratories, has been granted final approval from the United States Food and Drug Administration (USFDA) for its Abbreviated New Drug Application (ANDA) Milrinone Lactate in 5% Dextrose Injection, 20 mg/100 mL (0.2 mg/mL) and 40 mg/200 mL (0.2 mg/mL) Single-dose Infusion Bags, a generic therapeutic equivalent version of the Reference Listed Drug (RLD) PRIMACOR in Dextrose 5%, by Sanofi Aventis US LLC.
Milrinone is indicated for the short-term intravenous treatment of patients with acute decompensated heart failure. According to IQVIATM (IMS Health), Milrinone Lactate in 5% Dextrose Injection had US sales of approximately $11 million for the 12-month period ending July 2025.

4 mins read . Sep 5, 2025
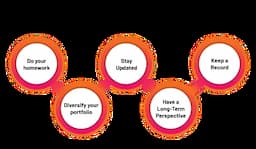
3 mins read . Aug 20, 2025

8 mins read . Aug 18, 2025
Recent Article
- •
- •
- •
- •